BIR572 -- Regulatory Mechanisms of Eukaryotic Cells
Notes for Lectures on Yeast Genetics
March 20 -- March 27, 2007
This page was last updated March 20, 2007
Joel Huberman
Dept. of Cancer Biology
C&V 501
Extension 3047
Preferred e-mail address: huberman@buffalo.edu
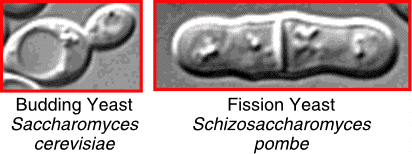
Saccharomyces cerevisiae and Schizosaccharomyces
pombe represent two very different types of yeast. Molecular evidence
suggests that they diverged over 1 billion years ago. S. cerevisiae
has "evolved" more rapidly than S. pombe since divergence.
In some ways, S. pombe resembles animal cells more than does S. cerevisiae (e.g. division pattern; centromeres; introns; heterochromatin), but in other respects S. cerevisiae is more similar to animal cells (e.g. relative lengths of G1 and G2; synaptonemal complexes).
Reasons why yeasts are excellent eukaryotic model organisms
- Easily manipulated in laboratory
- Rapid growth (1.5 or 2.5 hours)
- Non-pathogenic
- Stable diploid (cerevisiae) and haploid (both) states
- Easy mating of haploid cells to generate diploids
- Simple complementation tests
- Efficient transformation by exogenous DNA
- Integration
- Autonomously replicating plasmids
- Efficient homologous recombination
- Simple targeting of mutations into the genome
- Simple genetic screens
Fig. 1. Size and appearance of cells
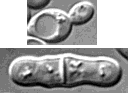
- S. cerevisiae ellipsoidal; about 5 µm diameter
- S. pombe cylindrical; 3-4 µm diameter; 7-15 µm long
- Divides by medial fission
Life cycles
- S. cerevisiae (Fig. 2)
exists in haploid and diploid state with two haploid mating types (a
and alpha)
- S. pombe (Fig. 3) exists
primarily in haploid state (with two mating types, h+ and h-) but enters
a transient diploid state as a result of mating. Under normal conditions,
diploid cells quickly begin meiosis and generate haploid spores.
- Notice in Fig. 3 that S. pombe asci are linear. In contrast, S. cerevisiae asci (not pictured in Fig. 2) are pyramidal. The pyramidal S. cerevisiae asci do not retain information regarding the order of meiotic events. However, the linear S. pombe asci do retain such information, as shown in Fig. 3b.
- Mating type switching: homothallism vs. heterothallism (Fig.
4)
- Studies of mating type switching in both yeasts have led to fascinating
results
- Frequent switching in wild type strains
- Heterochromatinization
- Fig. 4A
Cell cycles
Fig. 5
Metabolism
- Both organisms grow fastest on glucose, which is fermented into ethanol
or oxidized to CO2 and H2O
- S. cerevisiae prefers fermentation and will use it so long as
glucose is present, even in the presence of oxygen; S. pombe employs
pure fermentation only in the absence of oxygen
- S. cerevisiae shifts to aerobic growth on EtOH when the supply
of glucose runs out (diauxic shift)
Genomes
- Both yeasts
- DNA ~60% A+T
- Normal core histones; no close relative to histone H1
- Transcriptional silencing at silent mating type cassettes, telomeres, and rDNA
- S. cerevisiae
- 16 chromosomes
- Entire genome sequenced (12 Mb; about 6,000 genes)
- Centromeres small (~120 bp), unique; three regions bind proteins, allowing
interaction with a single microtubule for segregation; no transcriptional
silencing at centromeres
- Telomeres (TG1-3)n [Note
that human telomere sequence is (TTAGGG)n]
- Replication origins (ARS elements) 100-150 bp
- S. pombe
- 3 very large chromosomes (most markers on same chromosome unlinked
to each other)
- Entire genome sequenced (14 Mb; about 5,000 genes)
- Centromeres large (40-100 kb), mostly repetitious; may bind more than
a single microtubule; genes within centromere are usually
silenced
- Telomeres seem complicated at first: (T1-3AC0-1A0-2C0-1G1-8)n, but by far the most common repeat is TTACAGG [Note that human telomere sequence is (TTAGGG)n]
- Replication origins (ARS elements) 500-1500 bp
Extrachromosomal elements
- Mitochondria
- The fact that budding yeast cells can grow aerobically or anaerobically means
that mitochondrial respiratory function is not essential. For this reason,
mitochondrial DNA can be mutated at will, and mitochondrial genetics can
be readily studied. Note that although it is possible to delete the entire
mitochondrial genome, mitochrondrial membranes (which are nuclear encoded)
are essential for viability under all conditions and cannot be deleted.
- Mitochondrial mutants (S. cerevisiae) are recognized as mutants
able to grow (albeit poorly) on glucose (which can be fermented) but not
on glycerol (which can only be metabolized by aerobic respiration).
- Fission yeast mitochondrial DNA is essential for wild-type cells, but mutations in certain nuclear genes (I don't believe these genes are characterized yet) permit isolation of fission yeast cells lacking mitochondrial DNA.
- Plasmids
- S. cerevisiae
- 2 µm circle
- 6.3 kbp
- 60-100 copies/cell
- S. pombe - no known naturally occurring plasmids
- Non-Mendelian determinants in S. cerevisiae
- Prion-like proteins
- Dominant
- Extranuclear
- Not associated with killer particles or mitochondria
- Self-seeding
- Examples:
- [PSI+]/[psi-]
- Increases the level of frameshift and ochre suppression
- Altered structure (aggregated) form of Sup35p, which is a polypeptide chain release factor
- [URE3]/[ure3-]
- Affects nitrogen metabolism
- Altered structure (aggregated) form of Ure2p, which is a regulator of nitrogen catabolism
- Killer particles
- Defective dsRNA virus encoding a lethal toxin
- Studied by Jeremy Bruenn at UB (among others)
Genetic nomenclature
- S. cerevisiae
- Gene names are 3 letters followed by a number
- Dominant alleles are capitalized; recessives are lower case; usually
but not always the wild-type is dominant
- Gene names are italicized (YFG1)
- Wild type may, optionally, be denoted by a + sign (YFG1+)
- Mutant alleles are indicated by an allele-specific number (yfg1-1)
- Knockouts are indicated yfg1::URA3 or yfg1Δ
- Proteins are partially lower case, not italicized: Yfg1 or Yfg1p
- S. pombe
- Gene names are 3 lower case letters followed by a number
- Wild type is denoted by a + sign (yfg1+)
- Mutants are denoted by an allele number (yfg1-1)
- Knockouts are designated yfg1::ura4 or yfg1Δ
Making mutants
- Ethyl methane sulfonate (safer; base transitions); nitrosoguanidine
(more effective; base transitions); UV (very safe, least effective, many
different types of mutation)
- 5 x 10-4 to 1 x 10-2 per gene, with 20-80% killing
- Screen for desired phenotype
- Backcross desired mutants to wild-type
- Tetrad analysis to confirm single mutant status
- Mate; sporulate; dissect asci; determine phenotypes of spore colonies
- Fig. 6 (results predicted if single
gene)
- Can be used for mapping; see below
Random spore analysis (a poor or clumsy person's alternative to tetrad analysis)
- Mass isolation of spores from asci followed by characterization of
phenotypes of the resulting spore colonies permits much larger numbers
of spores to be analyzed without the difficulties of micromanipulation,
but ascus information is lost, and dead spores can't be identified
- If mutation is single gene, then half of spore colonies (from cross
of mutant x wt) will show mutant phenotype
- If none of the phenotypes is lethal, then analysis of random spores
can also provide recombination frequencies, which can be used for mapping
- S. cerevisiae
- Diploids of desired genotype are induced to sporulate on starvation
medium
- Potential problem due to incomplete sporulation; some vegetative diploid
cells inevitably remain
- One approach is to use a can1-r allele in one of the haploid
parent strains
- Diploid cells (CAN1-S+/can1-r) and half the spores (CAN1-S+)
will be killed by canavanine; the survivors will all be spore colonies
- This procedure works generally, except for the rare cases in which one of the genes of interest is linked to the CAN1 gene
- After 2 days on starvation medium, incubate briefly with Glusulase (an enzyme prep from snails that hydrolyzes yeast cell walls) and shake with glass beads to remove the ascus wall and free the spores
- Plate onto appropriate medium containing canavanine (and other supplements,
as appropriate)
- Check phenotypes of spore colonies by appropriate methods
- S. pombe
- Generation of free spores is facilitated by tendency of diploids to spontaneously sporulate and
by fragility of asci and vegetative cells
- Make diploids from desired haploids by crossing on ME (malt extract;
carbohydrate only; no nitrogen) plates. Incubate 2 days at 25°.
- Absence of nitrogen leads to G1 arrest and promotes mating competence.
Like S. cerevisiae cells, S. pombe cells mate while in G1.
- Collect some of mating mixture (which will contain asci due to spontaneous
sporulation of diploids) and incubate briefly with Glusulase.
- Opens asci, liberates spores, kills haploid and diploid vegetative
cells
- Plate on appropriate medium to generate spore colonies
- Check phenotypes of spore colonies
Complementation
- Used to test whether two mutations producing the same phenotype are
in the same or different "complementation groups" (Fig. 7)
- Procedure in S. cerevisiae (Fig. 8)
- Introduce desired mutations into a and alpha strains
with complementary nutritional markers
- Plate a and alpha strains to be tested in parallel streaks
on YPD plates, one plate for a strains and one for alpha.
Incubate 24 hrs, 30°.
- Replicate a and alpha plates onto velveteen at right
angles. Transfer to fresh YPD plate. Incubate 24 hrs, 30°, to allow
mating at intersections.
- Replicate onto SD plate. Incubate several days, 30°.
- Pick cells from single colonies, characterize for mutant phenotype
(can be combined with previous step).
- Procedure in S. pombe (Fig. 8b)
- Rendered more difficult by
- instability of diploid cells, which tend to sporulate spontaneously
- requirement for nutrient starvation before mating
- One work-around employs the mat2-102 strain, which mates with
h- cells to produce non-sporulating diploids. This strain was previously
known as the mei1-102 strain.
- An alternative work-around involves introducing desired mutations into
h+ and h- strains with ade6-M210 or ade6-M216 mutations,
which are capable of intra-genic complementation to permit growth in the
absence of adenine.
- Mix resulting strains in patches on ME plate (plus appropriate supplements).
Incubate at 25°.
- After 16-24 hrs (before diploids have had a chance to sporulate), streak
a loopful of each cross onto MM (minimal medium) containing a nitrogen
source and phloxine B. Incubate at 30° for 3 days.
- After 3 days, check colonies for staining intensity. Diploid colonies
should stain dark red. If haploid ade6-M210 and ade6-M216
strains were crossed, then diploids should be able to grow (due to intragenic complementation), but cells in
these viable diploid colonies will frequently attempt to sporulate, and
(rarely) haploid recombinants capable of vigorous, adenine-independent growth may be produced by intragenic
recombination. Such viable haploid colonies would stain more lightly (pink)
with Phloxine B.
- Check cells from single diploid colonies for phenotype of interest
(can be combined with previous two steps).
Mapping by tetrad analysis
- Possible outcomes of 2-factor cross (A/a and B/b)
- Genes on different chromosomes (Fig.
9)
- Parental ditype (PD) and nonparental ditype (NPD) equally probable
- If either A or B is so far from their respective centromeres
that the probability of recombination (crossover) between them and their
centromeres is 50%, then the probability of tetratypes (T) is 4 times that
of PD and NPD. You can convince yourself of this if you construct a 6 x
6 table in which the 6 possible arrangements of A and a in
the 4 spores (AAaa, AaAa, AaaA, aAAa, aAaA, aaAA) are listed on one side,
the 6 possible arrangements of B and b are listed on a second
side (at right angles to the first), and the genotypes of the spores with
these arrangements are listed within the table. You will find that PD:NPD:T
= 6:6:24 = 1:1:4.
- Crossover between A and its centromere causes A and a to stay together
at first meiotic division and not separate until second division. Called
"second division segregation" (SDS). Genes close to their centromeres
display reduced SDS. Note that SDS gives rise to tetratypes (T), so the
frequency of SDS is the frequency of T.
- Detecting reduced SDS requires that both genes in the cross be close to their centromeres; otherwise the two segregate totally randomly with respect to each other and the reduced SDS of one of them cannot be detected.
- You may find it difficult to understand why the same results would be obtained when both genes are far from their centromeres and when one gene is far but the other gene is close. You can assist your understanding if you draw out (make a figure showing) the consequences of single crossovers between both of the genes involved in the cross and their centromeres. You'll notice that when both genes are involved in crossovers, the effects of the two crossovers can cancel each other out, leading to PDs or NPDs rather than Ts. Thus when both genes are far from their centromeres, there's no increased frequency of Ts (compared to one gene close to and one gene far from its centromere), because some of the crossovers occur on both chromosomes simultaneously and don't produce Ts. See Fig. 10 for an illustration of what happens when one gene is extremely close to its centromere (no significant recombination between the gene and its centromere), and the other gene is far from its centromere.
- When both genes are close to their centromeres, then the frequency
of crossovers between both genes and their centromeres is reduced, the
frequency of Ts is reduced, and the ratio PD:NPD:T = 1:1:<4. This should
be obvious from Fig. 9, which shows
that crossovers between genes and their centromeres are required to produce
Ts.
- For most sensitive detection of centromere linkage of an unknown gene
(A), a cross should be made with a gene known to be so close to
its centromere that the probability of recombination between it and its
centromere is essentially 0. TRP1 on chromosome IV of S. cerevisiae
is a good example of such a tightly centromere-linked gene. Thus the extent
to which A is centromere-linked can be determined by tetrad analysis
of the cross (A TRP1) x (a trp1) or (A trp1)
x (a TRP1). See Fig. 10 for
further explanation.
- Genes on the same chromosome
- If there are no crossovers between the genes, then PDs are produced.
- Single crossovers produce Ts (Fig.
11)
- 1/4 of double crossovers produce NPDs (Fig.
12)
- In a recombinant ascus (T or NPD), 2 (half) of the strands recombine if there is a single crossover and 4 strands recombine if there is a double crossover. Thus, even if all of the strands do not participate in the double crossover (Fig. 12), other strands participate more than once so that the total number of participating strands is 4, which is equal to the number of spores in the ascus.
- If genes are close, so that map distance is less than or equal to 40
centimorgans (cM), then the following equations (from Perkins, D.D. Biochemical
mutants of the smut fungus Ustilago maydis. Genetics 34: 607-626, 1949)
can be used to calculate recombination frequency and map distance. These
equations do not account for more than two crossovers in an interval or
for chromatid interference (local deviations from this formula).
- Recombination frequency = total recombination events/total spores
- = total recombination events/4(total asci)
- = (2[single crossovers] + 4[double crossovers])/4(total asci)
- = (1/2[single crossovers] + double crossovers)/total asci
- = (1/2[T-2NPD] + 4NPD)/(PD + NPD + T)
- = (T + 6NPD)/2(PD + NPD + T)
- Note: the frequency of all double crossovers = 4NPD. Some of the Ts
are also due to double crossovers. The frequency of Ts due to double crossovers
is 2NPD (Fig. 12). Thus T-2NPD is
the frequency of single crossovers.
- Multiply by 100 to get map distance in centimorgans
Modern mapping procedures
Other modern techniques (just a few examples from many possibilities)
- Introducing DNA on plasmids (Fig.
13)
- Integrating vectors
- ARS vectors
- CEN vectors (not needed for S. pombe)
- Cloning by complementation
- Simplest when mutant phenotype is recessive and can be selected against
- Also easy when mutant phenotype is dominant and can be selected for
- Straightforward
- Transfect genomic or cDNA library on ARS plasmid into mutant cells
(mutation in yfg)
- Select for wild-type phenotype
- Rescue plasmid bearing suppressor gene into E. coli
- Sequence insert
- Test to make sure the cloned suppressor gene maps at the position of
the mutant yfg gene; that is, have you cloned the gene of interest
or a second-site suppressor?
- Check by locating sequence of cloned suppressor in genome database
that cloned suppressor gene is located in same portion of genome as YFG,
OR
- Integrate selectable marker into genome at position of cloned suppressor
gene
- Cross to cells bearing mutant yfg
- Tetrad analysis will display 2:2 segregation (100% PD; no recombination)
of selectable marker and yfg if cloned suppressor gene is YFG
- Gene replacement
- One-step (Fig. 14)
- Useful for gene knockouts
- Can be used to insert linear PCR products, if they contain selectable markers and terminal regions of homology to the yeast genome
- Two-step (Fig. 15)
- Useful for introducing any mutation into the chromosome at any location
- Two-hybrid (Fig. 16)
- Stanley Fields, Roger Brent
- GAL4 or LexA-BD-bait
- GAL4 or synthetic-AD-prey
- Indicator proteins (His3, Leu2, lacZ)
- Uses
- Find prey proteins in library
- Identify regions important for interaction in either bait or prey
- DNA microarray (Fig. 17)
- Generate stretches of DNA representing entire genome (for example,
gene sequences)
- Apply in array to microscope slide
- Hybridize with fluorescent (c)DNA probe of interest and simultaneously
with differently colored fluorescent control probe
- Scan and analyze
- Genome-wide mutant libraries--examples
- Bar-coded gene deletions (Fig. 18)
- Synthetic Genetic Array analysis (Fig. 19)
- Identifies synthetic lethal and synthetic sick interactions on a genome-wide scale
Here are some review aids. Clicking on the links below will cause a Microsoft Word Document to be downloaded to your hard drive, or to be opened in Word on your computer, depending on the settings in your web browser.
Exam questions from 2001
Exam answers from 2001
Exam questions from 2002
Exam answers from 2002
Exam questions from 2003
Exam answers from 2003
Exam questions from 2004
Exam answers from 2004
Exam questions from 2005
Exam answers from 2005
Exam questions from 2006
Exam answers from 2006
References (where to find more)
Web References
Books
- Broach, J.R., Pringle, J.R. and Jones, E.W., Editors. The Molecular and Cellular Biology of the Yeast Saccharomyces: Genome Dynamics, Protein Synthesis and Energetics. Cold Spring Harbor Laboratory Press, Cold pring Harbor, New York, 1991.
- Guthrie, C. and Fink, G.R., Editors. Guide to Yeast Genetics and Molecular Biology. Methods in Enzymology, Volume 194, Academic Press, New York, 1991.
- Broach, J.R., Pringle, J.R. and Jones, E.W., Editors. The Molecular and Cellular Biology of the Yeast Saccharomyces: Gene Expression. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1992.
- Alfa, C., Fantes, P., Hyams, J., McLeod, M., Warbrick, E. Experiments with Fission Yeast: A Laboratory Course Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1993.
- Hall, M.N. and Linder, P., Editors. The Early Days of Yeast Genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1993.
- Adams, A., Gottschling, D. E., Kaiser, C. A., and Stearns, T. Methods in Yeast Genetics: A Cold Spring Harbor Laboratory Course Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1997.
- Broach, J.R., Pringle, J.R. and Jones, E.W., Editors. The Molecular and Cellular Biology of the Yeast Saccharomyces: Cell Cycle and Cell Biology. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1997.
- Burke, D., Dawson, D., Stearns, T. Methods in Yeast Genetics: 2000 Edition. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 2000.
- Fantes, P. and Beggs, J., Editors. The Yeast Nucleus (Frontiers in Molecular Biology, 33) Oxford University Press, 2000.
- Egel, R., Editor. The Molecular Biology of Schizosaccharomyces pombe: Genetics, Genomics and Beyond. Springer-Verlag, Berlin, 2004.
- Gould, K., Editor. Protocols for Experimentation with Schizosaccharomyces pombe. Methods 33(3): 187-263, 2004.
Papers for class discussion
- Hartwell, L.H., Mortimer, R.K., Culotti, J. and Culotti, M. Genetic control of the cell division cycle in yeast: V. Genetic analysis of cdc mutants. Genetics 74: 267-286, 1973.
- Tong AHY, Evangelista M, Parsons AB, Xu H, Bader GD, Page N, Robinson M, Raghibizadeh S, Hogue CWV, Bussey H, Andrews B, Tyers M, Boone C. Systematic genetic analysis with ordered arrays of yeast deletion mutants. Science 294: 2364-2368, 2001.
Supplementary papers (and papers assigned for discussion in previous classes)
- Hereford, L.M. and Hartwell, L.H. Sequential gene function in the initiation of Saccharomyces cerevisiae DNA synthesis. J. Mol. Biol. 84: 445-461, 1974.
- Nurse, P., Thuriaux, P., and Nasmyth, K. Genetic control of the cell division cycle in the fission yeast Schizosaccharomyces pombe. Mol. Gen. Genet. 146: 167-178, 1976.
- Hicks, J., Strathern, J.N. and Klar, A.J.S. Transposable mating type genes in Saccharomyces cerevisiae. Nature 282: 478-483, 1979.
- Nasmyth, K. and Nurse, P. Cell division cycle mutants altered in DNA replication and mitosis in the fission yeast Schizosaccharomyces pombe. Mol. Gen. Genet. 182: 119-124, 1981.
- Hartwell, L.H. Twenty-Five Years of Cell Cycle Genetics. Genetics 129: 975-980, 1991.
- Chien, C.-T., Bartel, P.L., Sternglanz, R. and Fields, S. The two-hybrid system: a method to idnetify and clone genes for proteins that interact with a protein of interest. Proc. Natl. Acad. Sci. USA 88: 9578-9582, 1991.
- Guarente, L. Synthetic enhancement in gene interaction: a genetic tool comes of age. Trends in Genetics 9: 362-366, 1993.
- Fromont-Racine, M., Rain, J.-C., and Legrain, P. Toward a functional analysis of the yeast genome through exhaustive two-hybrid screens. Nature Genetics 16: 277-282, 1997.
- DeRisi, J.L., Iyer, V.R. and Brown, P.O. Exploring the metabolic and genetic control of gene expression on a genomic scale. Science 278: 680-686, 1997.
- Cho, R. J., Campbell, M. J., Winzeler, E. A., Steinmetz, L., Conway, A., Wodicka, L., Wolfsberg, T. G., Gabrielian, A. E., Landsman, D., Lockhart, D. J. and Davis, R. W. A genome-wide transcriptional analysis of the mitotic cell cycle. Mol. Cell 2: 65-73, 1998. See also http://genomics.stanford.edu.
- Basrai MA, Velculescu VE, Kinzler KW, Hieter P. NORF5/HUG1 is a component of the MEC1-mediated checkpoint response to DNA damage and replication arrest in Saccharomyces cerevisiae. Mol. Cell. Biol. 19: 7041-7049, 1999.
- Matsuura A, Naito T, Ishikawa F. Genetic control of telomere integrity in Schizosaccharomyces pombe: rad3(+) and tel1(+) are parts of two regulatory networks independent of the downstream protein kinases chk1(+) and cds1(+). Genetics 152: 1501-1512, 1999.
- Kiely J, Haase SB, Russell P, Leatherwood J. Functions of Fission Yeast Orp2 in DNA Replication and Checkpoint Control. Genetics 154: 599-607, 2000.
- Nasmyth K (2001) A prize for proliferation. Cell 107:689-701.
- Hall IM, Noma KI, Grewal SIS. RNA interference machinery regulates chromosome dynamics during mitosis and meiosis in fission yeast. PNAS 100:193-198 2003.
- Huang, D. and Koshland, D. Chromosome integrity in Saccharomyces cerevisiae: the interplay of DNA replication initiation factors, elongation factors, and origins. Genes Dev. 17:1741-1754, 2003.
- Tong, A. et al. Global mapping of the yeast genetic interaction network. Science 303:808-813, 2004.
- Rustici, G. et al. Periodic gene expression program of the fission yeast cell cycle. Nature Genetics 36:809-817, 2004.
- Thon G, Hansen KR, Altes SP, Sidhu D, Singh G, Verhein-Hansen J, Bonaduce MJ, Klar AJS. The Clr7 and Clr8 directionality factors and the Pcu4 cullin mediate heterochromatin formation in the fission yeast Schizosaccharomyces pombe. Genetics 171: 1583-1595, 2005.


